BIMZELX is indicated for the treatment of: moderate to severe plaque psoriasis (PsO) in adults who are candidates for systemic therapy; active psoriatic arthritis (PsA), alone or in combination with methotrexate, in adults who have had an inadequate response, or who have been intolerant, to one or more DMARDs; active non‑radiographic axial spondyloarthritis (nr-axSpA) with objective signs of inflammation as indicated by elevated CRP and/or MRI, in adults who have responded inadequately, or are intolerant, to NSAIDs; active axial spondyloarthritis (AS) in adults who have responded inadequately or are intolerant to conventional therapy; and active moderate to severe hidradenitis suppurativa (HS) in adults with an inadequate response to conventional systemic HS therapy.
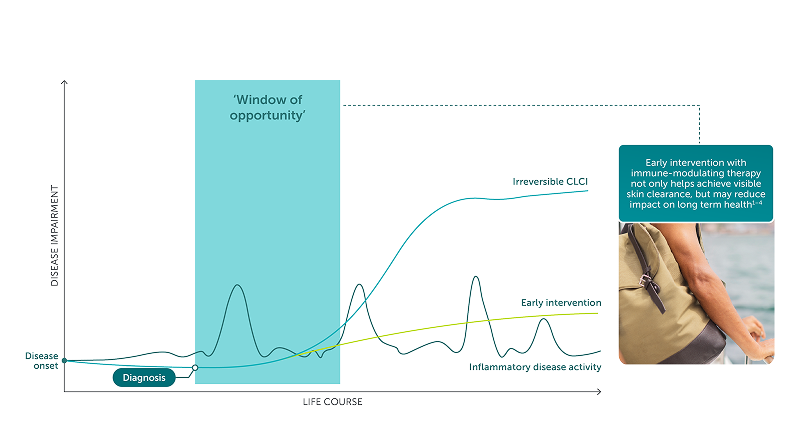
Rising To The Challenge of Psoriatic Disease
ACT Now With BIMZELX
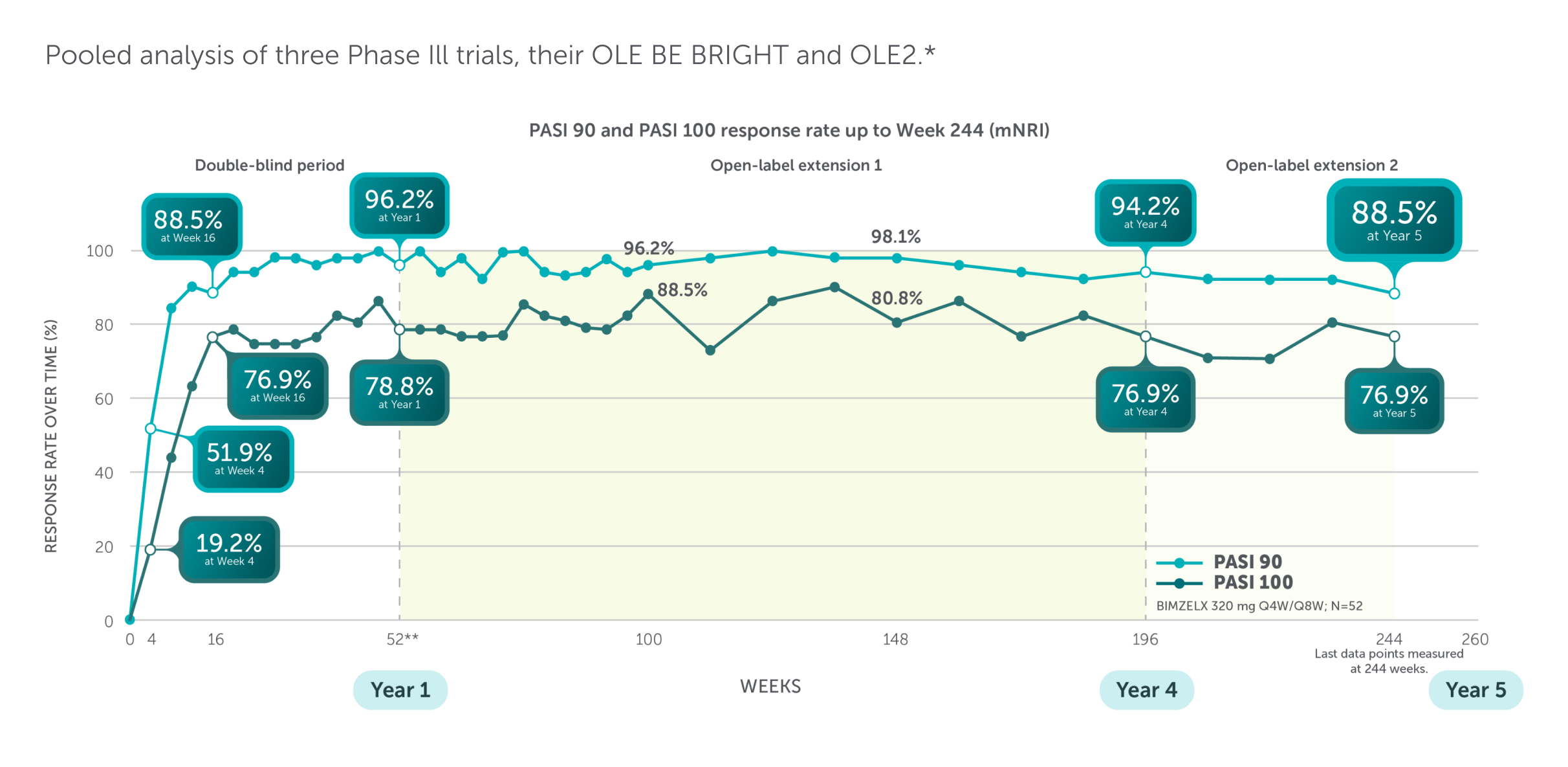
Emerging Data
PHASE III/IIIb
BIMZELX demonstrated improved skin efficacy vs three other biologic therapies1–4
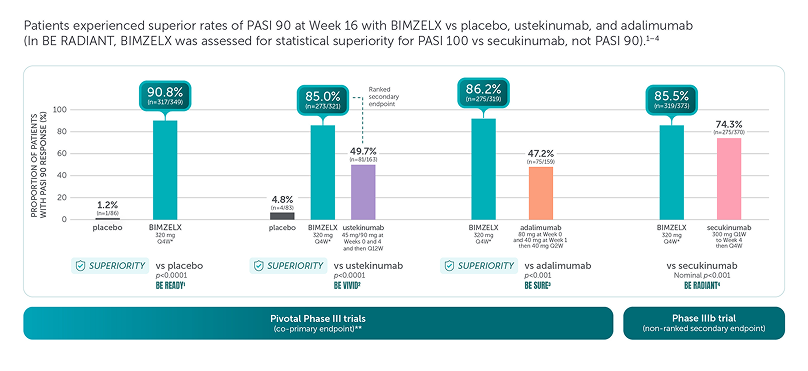
These are four separate studies and should not be directly compared. Adapted from references 1–4.
Missing data were imputed with non-responder imputation; p values for the comparison of treatment groups were based on CMH test from the general association.1–4
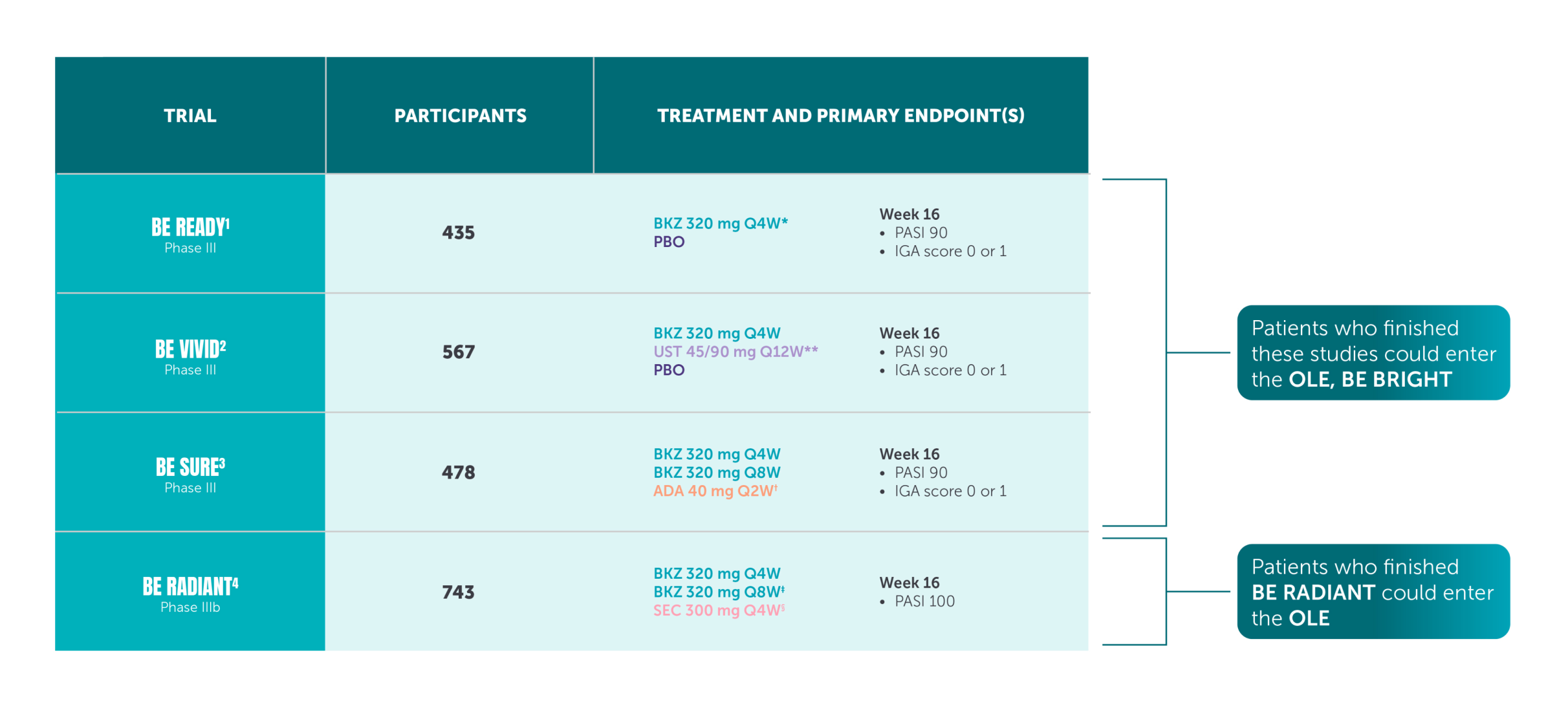
*The recommended dose for adult patients with plaque PsO is 320 mg (given as two subcutaneous injections of 160 mg or one subcutaneous injection of 320 mg) at Week 0, 4, 8, 12, 16 and every 8 weeks thereafter.5
**The co-primary efficacy endpoints for the pivotal phase III studies BE READY, BE VIVID, and BE SURE were PASI 90 and IGA score of clear or almost clear (IGA 0/1) at Week 16.1–4 †The primary endpoint of BE RADIANT was PASI 100 response at Week 16 with BIMZELX vs secukinumab.4
aRD, adjudicated risk difference; CI, confidence interval; CMH, Cochran-Mantel-Haenszel; PASI 90, ≥90% improvement from baseline in Psoriasis Area and Severity Index; Q1W, once every week; Q2W, every 2 weeks; Q4W, every 4 weeks; Q12W, every 12 weeks.
References: 1. Gordon KB, et al. Lancet. 2021;397:475–486. 2. Reich K, et al. Lancet. 2021;397:487–498. 3. Warren RB, et al. N Engl J Med. 2021;385:130–141. 4. Reich K, et al. N Engl J Med. 2021;385:142–152. 5. BIMZELX SmPC.
Patients receiving BIMZELX had visibly improved skin clearance at 16 weeks1–4
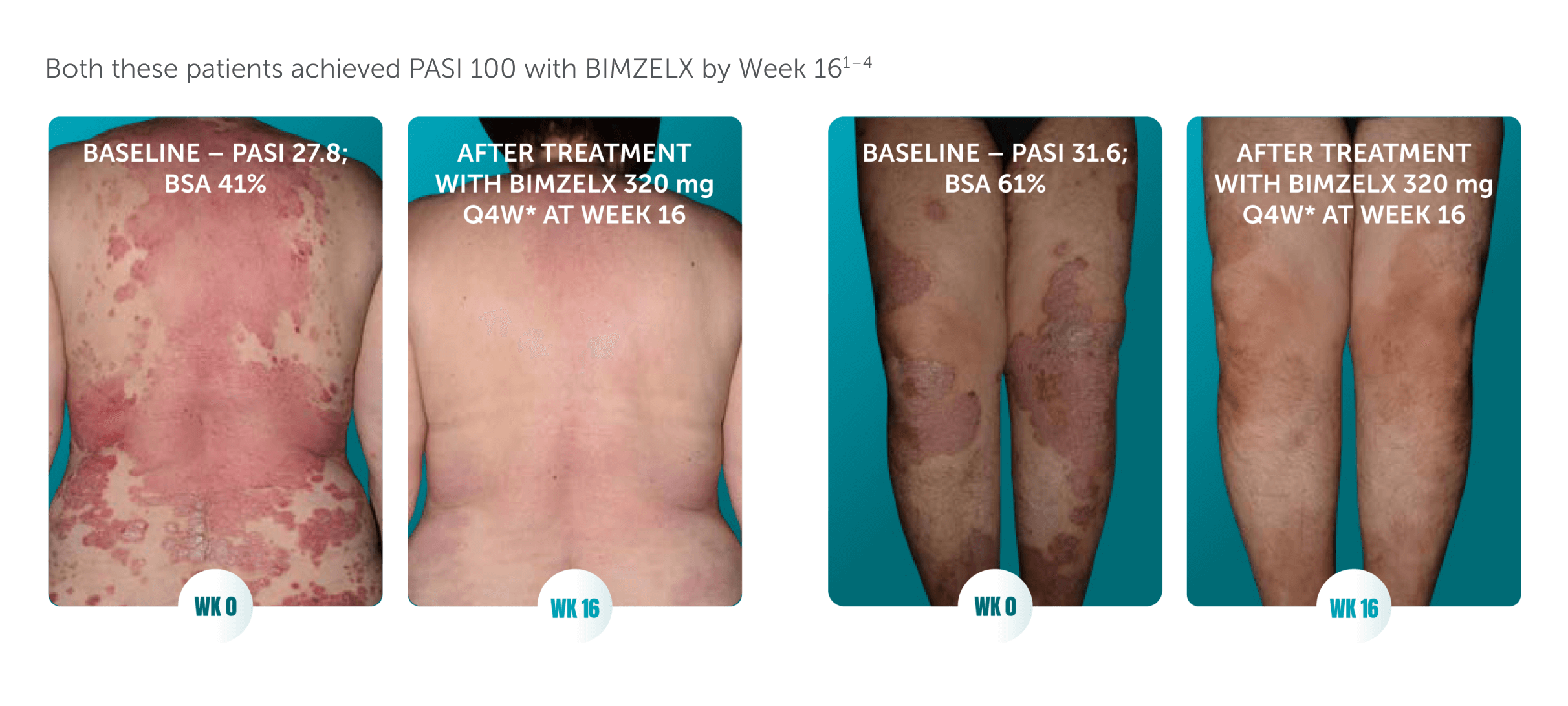
Actual clinical trial patients from BE RADIANT (left)1,2 and BE VIVID (right).3,4 The patients consented to having their photograph taken and used in promotional materials. These are demonstrations of individual patient’s responses. Individual results may vary.
BSA, body surface area; PASI 100, 100% improvement from baseline in Psoriasis Area and Severity Index; Q4W, every 4 weeks; WK, week.
References: 1. Reich K, et al. N Engl J Med. 2021;385:142–152. 2. UCB Data on file. 2020 BE RADIANT – patient photos. 3. Reich K, et al. Lancet. 2021;397:487–498. 4. UCB Data on file. 2020 BE VIVID – patient photos. 5. BIMZELX SmPC.
BIMZELX demonstrated high rates of complete clearance for high-impact sites up to 4 years1
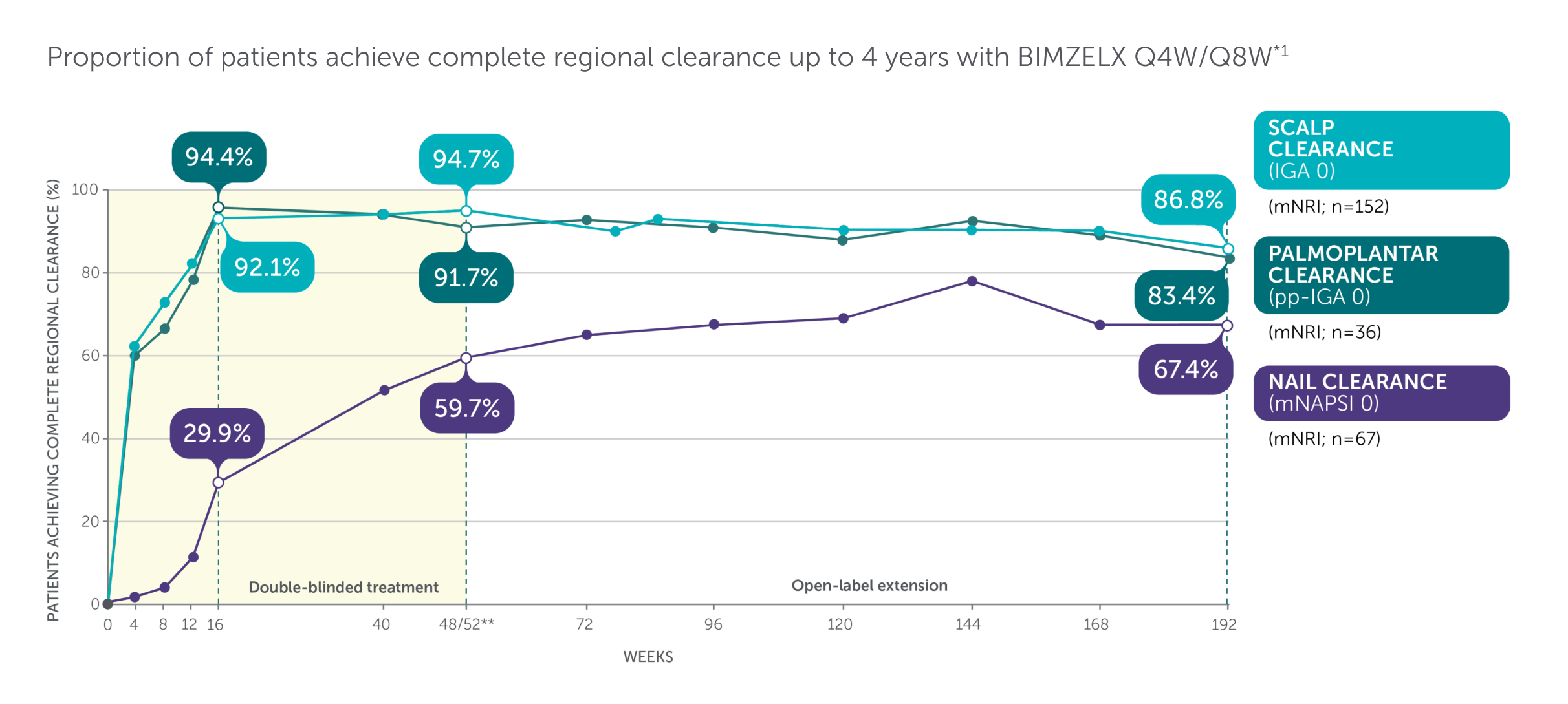
Adapted from Merola 2024.1
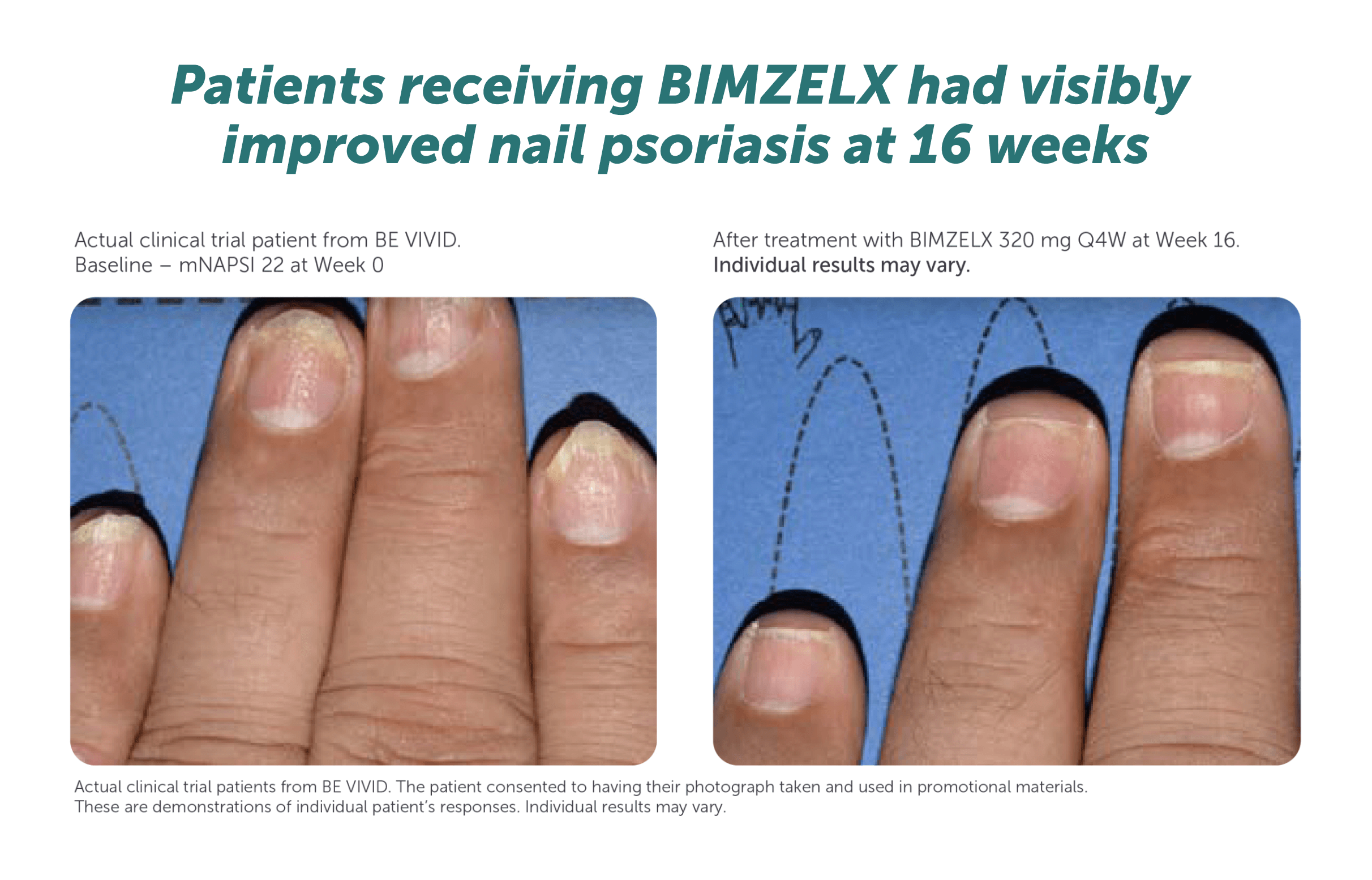
Includes patients with IGA ≥3. mNAPSI >10, or pp-IGA ≥3 at baseline. *Data are reported using modified non-responder imputation (mNRI): patients who discontinued due to lack of efficacy or treatment-related adverse events were considered non-responders; multiple imputation was used for other missing data. mNRI for scalp clearance, n=152. mNRI for nail clearance, n=67. mNRI for palmoplantar clearance, n=36. **Week 48/52 data are from Week 48 of BE SURE and BE READY, and Week 52 of BE VIVID, due to differences in assessment schedules.1 Post-hoc analysis of data pooled from BE VIVID (52 weeks), BE READY and BE SURE (56 weeks), and 3 years of their OLE, BE BRIGHT.1 BIMZELX Q4W/Q8W patients received BIMZELX 320 mg Q4W to Week 16, then BIMZELX Q8W throughout the maintenance period and on OLE entry.1 Scalp IGA was a listed original secondary endpoint for BE READY and BE VIVID.2,3 Change in mNAPSI score and pp-IGA response (in patients with nail and palmoplantar psoriasis at baseline) were exploratory endpoints in BE READY and BE VIVID.4,5
IGA, Investigator’s Global Assessment; mNAPSI, modified Nail Psoriasis Severity Index; mNRI, modified non-responder imputation; OLE, open-label extension; pp, palmoplantar psoriasis; Q4W, every 4 weeks; Q8W, every 8 weeks.
References: 1. Merola JF. EADV 2024; P3320. 2. Gordon KB, et al. Lancet. 2021;397(10273):475–486. 3. Reich K, et al. Lancet. 2021;397:487–498. 4. Gordon KB, et al. Lancet. 2021;397(10273):475–486. Supp App. 5. Reich K, et al. Lancet. 2021;397:487–498. Supp App.
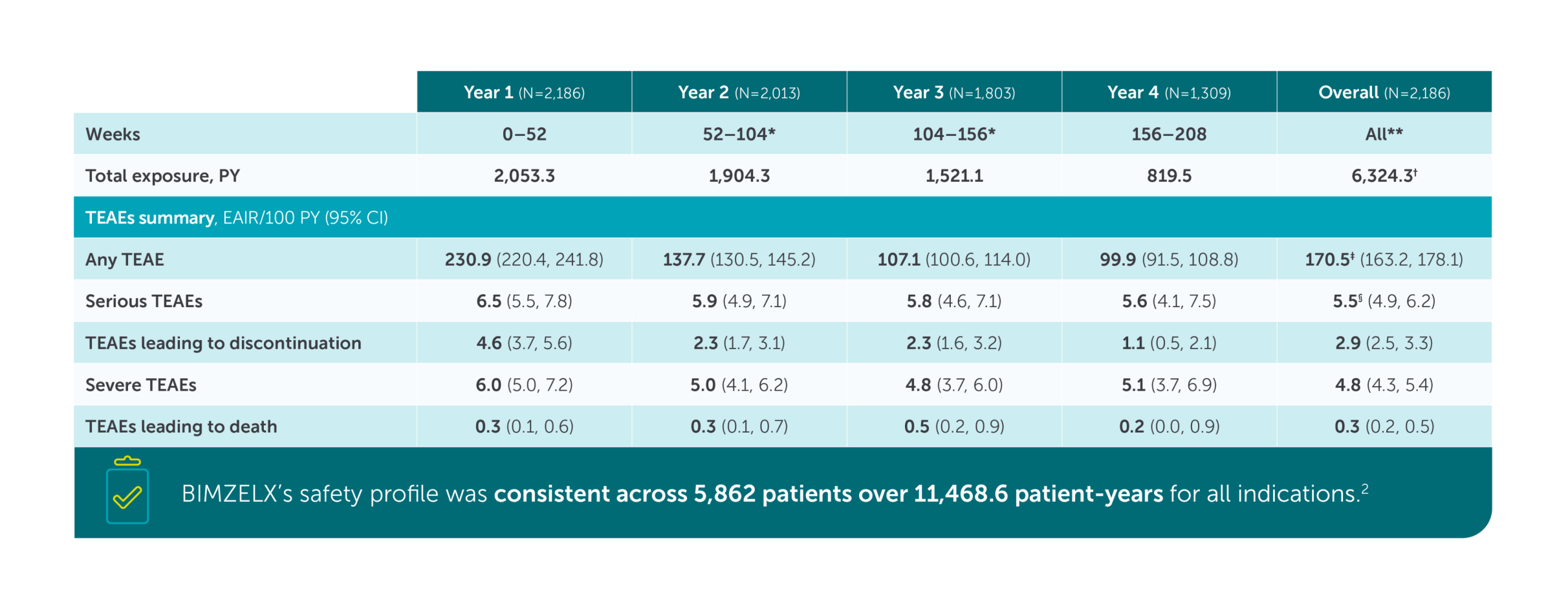
Safety Profile
Prescribing
Study details
Prescribing Information for Healthcare Professionals in the United Kingdom, click here
▼ This medicine is subject to additional monitoring. This will allow quick identification of new safety information. Adverse events should be reported. Reporting forms and information can be found at https://yellowcard.mhra.gov.uk for United Kingdom and www.hpra.ie/homepage/about-us/report-an-issue for Republic of Ireland. Adverse events should also be reported to UCB Pharma Ltd at UCBCares.UK@UCB.com or 0800 2793177 for the UK and UCB (Pharma) Ireland Ltd at UCBCares.IE@UCB.com or 1800 930075 for Republic of Ireland
Prescribing Information for Healthcare Professionals in the Republic of Ireland
Prescribing Information for HCP's in Republic of Ireland
(Please consult the Summary of Product Characteristics (SmPC) before prescribing)